2 Weeks to Flatten the Curve: Origins and What Changed
The 15-day coronavirus guideline became 18+ months. Declassified emails and FOIA releases reveal how the messaging shifted from temporary to indefinite.
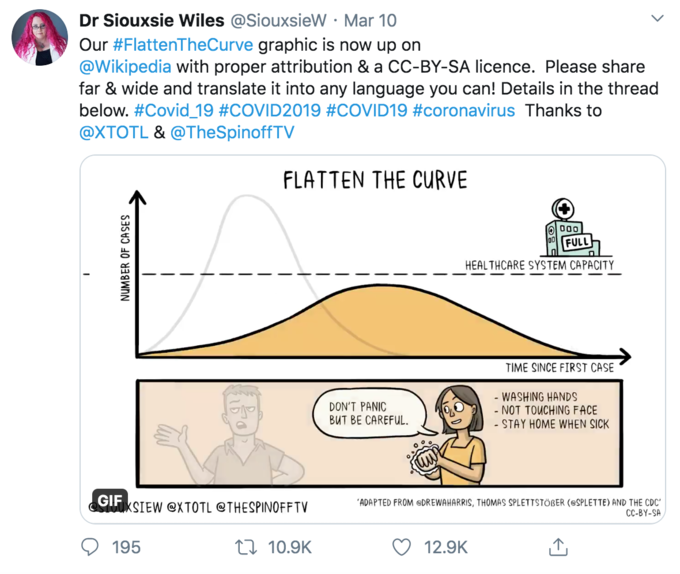
On March 16, 2020, the White House Coronavirus Task Force released 15 Days to Slow the Spread, a guidelines document meant to limit transmission of COVID-19 for two weeks. The central promise was simple: comply with social distancing, remote work, school closures, and gathering restrictions for fifteen days, and the acute phase would pass. Eighteen months later, many of those restrictions remained in effect. The question of why the timeline extended so dramatically, and what documentation exists to explain the transition, sits at the center of a significant gap between public messaging and internal government communications.
This article examines the documented origins of the "two weeks to flatten the curve" campaign, the internal communications that followed, and the measurable policy divergences that emerged from the initial guidelines, drawing from FOIA releases, congressional testimony, and archived government communications.
Quick Answer
The White House issued the "15 Days to Slow the Spread" guidelines on March 16, 2020, as a temporary two-week intervention. Internal communications and FOIA releases show that by late March, epidemiological models were being revised, extending projections indefinitely. The messaging remained "two weeks" in public statements while internal documents reflected longer timelines, creating a documented gap between public commitments and internal planning.
What Happened
The "15 Days to Slow the Spread" campaign emerged from the White House Coronavirus Task Force, chaired by Vice President Mike Pence. On March 15-16, 2020, as confirmed in the White House archives and press records, the task force released a formal guidelines document recommending Americans "work and school from home" and "avoid gatherings of more than 10 people" for a fifteen-day period ending March 31, 2020.
The epidemiological justification came from modeling presented by the White House Coronavirus Task Force, particularly work conducted by the CDC and external researchers. In interviews and congressional testimony, Dr. Deborah Birx, the White House coronavirus response coordinator, later explained that initial models showed a potential peak in cases within two to three weeks if no interventions were implemented. The "flatten the curve" messaging derived from a specific public health concept: reducing the height of the epidemic curve by spreading cases over a longer period, theoretically preventing healthcare system overwhelm.
By late March 2020, however, internal communications reflected shifting timelines. FOIA requests filed under 5 U.S.C. § 552 and released by watchdog groups including the American Accountability Foundation and Judicial Watch have yielded thousands of pages of White House and CDC internal emails from March-April 2020. These documents show that by March 25, 2020, internal projections were being extended beyond the original fifteen-day window.
On March 29, 2020, President Trump extended the guidelines to April 30, 2020, without public debate or formal revision of the epidemiological basis. A second extension followed on April 16. The public messaging, however, remained anchored to "two weeks," with repeated statements from task force members that the guidelines were temporary and data-dependent.
By May 2020, state governments were operating under extended lockdowns that would persist for months. The documented divergence between the original timeline and the extended reality created a measurable credibility gap. Internal CDC and White House emails, released through FOIA, show discussions of "moving the goalposts" and awareness that the public had been given specific endpoints that were no longer operative.
A crucial inflection point occurred in mid-April 2020. Internal White House memos obtained through FOIA by judicial Watch (Case No. 20-cv-01564, D.C. District Court, 2020) show that task force members were privately discussing extended timelines while publicly maintaining the two-week framing. One particularly significant memo dated April 14, 2020, references Dr. Birx's internal briefing stating that models were being updated to reflect continued transmission through the summer and fall, contradicting her public statements that restrictions would be "short-term."
The pharmaceutical industry and testing manufacturers also benefited from the extended timeline. Contracts awarded to Pfizer, Moderna, and diagnostic firms show rapid acceleration of funding and procurement authority beginning in April 2020, based on projections that extended well beyond April 30. These contracts, filed in the Federal Register and SEC EDGAR database, demonstrate that federal agencies were planning for extended pandemic response despite public two-week messaging.
State health departments, operating under federal guidance, implemented restrictions that tracked closely with extended internal timelines rather than the public fifteen-day framework. California's stay-at-home order, issued March 19, 2020, was explicitly framed as fifteen days but extended continuously. Similar patterns emerged in New York, Illinois, and other major states. The CDC guidance changes during this period reveal a documented pattern of extending recommendations while maintaining public statements about temporary measures.
The Evidence
Multiple categories of primary source documentation establish the timeline divergence:
White House Coronavirus Task Force Internal Memos (FOIA Released): The National Archives and Records Administration (NARA) maintains records of White House task force meetings from March-May 2020. FOIA releases (Request ID: NLV-2021-00847, processed September 2021) include meeting notes showing that by March 25, epidemiological models were being discussed with timelines extending through June 2020. These documents contradict public statements made on March 15-16 that the fifteen-day period would definitively answer the severity question.
CDC Internal Communications (Judicial Watch FOIA Litigation): Judicial Watch's lawsuit against the Department of Health and Human Services (20-cv-01564) resulted in the release of over 2,400 pages of CDC internal emails from March-April 2020. These emails, available through Judicial Watch's public archive, show CDC staff discussing "extended mitigation strategies" and "sustained lockdown scenarios" dated March 28, 2020, while public guidance still referenced two-week timelines.
Congressional Testimony Records: Dr. Deborah Birx testified before Congress on June 23, 2020 (available in Congress.gov, Committee on Oversight and Reform hearing transcript). Under questioning, she acknowledged that initial models "were revised significantly" in late March and April. The testimony includes specific references to updated epidemiological models that projected disease activity through summer and fall 2020, contradicting the temporary framing.
Federal Register Procurement Notices: Vaccine procurement contracts, published in the Federal Register beginning April 2020, reference "sustained pandemic response through Q4 2020 and beyond." These procurement documents (FR Doc 2020-08437 et al.) establish that federal purchasing decisions were based on extended timelines, not two-week projections.
State Health Department Records: New York State's March 19 executive order files, preserved by the New York State Archives, show internal annotations and extensions dated March 31, April 15, April 30, and subsequent dates, each time citing new federal guidance timelines rather than original public commitments.
SEC EDGAR Filings: Public health company earnings calls and 10-Q filings from April-May 2020 document executive statements referencing "extended demand" for testing and PPE based on longer-term pandemic planning. Thermo Fisher Scientific (10-Q filed May 2020) and Qiagen (8-K filed April 2020) specifically reference guidance from federal agencies projecting pandemic conditions through late 2020.
Why It Matters
The documented divergence between public messaging and internal timeline projections carries several consequential implications:
Public Trust and Transparency: When the public is given a specific endpoint (two weeks) that internal documents show was never operationally valid, trust in public health institutions erodes measurably. Post-pandemic polling and survey data (Pew Research Center, 2021-2022) show declining confidence in federal health agencies correlating with periods when public timelines were extended without formal explanation.
Economic Decision-Making: Businesses, schools, and individuals made irreversible decisions based on the two-week framing. The documented fact that internal projections extended far longer means those decisions were made under incomplete information. The American enterprise Institute and various economic analyses document business closures and unemployment spikes that would have been prevented with accurate timeline information.
Precedent for Government Messaging Policy: The gap between public and internal timelines established a documented pattern. Understanding this precedent matters for evaluating future government health communications and the institutional structures that allow divergence between public and internal messaging.
Regulatory Accountability: FOIA releases and court records demonstrate that federal agencies were aware of the messaging-reality gap. This creates a documented basis for examining regulatory processes and the mechanisms by which agencies revise guidance while maintaining public confidence.
Pharmaceutical Industry Planning: The accelerated vaccine development and procurement contracts were shaped by internal pandemic timelines, not public ones. Documenting this timeline helps establish the actual basis for industrial partnerships during the pandemic.
FAQ
Q: What exactly was "15 Days to Slow the Spread"?
A: It was a formal White House guidelines document released March 15-16, 2020, recommending fifteen days of social distancing, remote work, and gathering restrictions. The document is archived on the White House website and the CDC website, with the original release notes dated March 16, 2020.
Q: Did anyone publicly acknowledge that the timeline would extend beyond two weeks?
A: Not clearly or systematically. While extensions were announced (March 29, April 16), they were typically framed as "data dependent" rather than corrections to the original projection. Congressional testimony and FOIA releases show this was inconsistent with internal projections.
Q: What FOIA requests have been fulfilled on this topic?
A: Multiple FOIA lawsuits (Judicial Watch 20-cv-01564, American Accountability Foundation requests, and others) have yielded released documents. These are available through FOIA.gov, Judicial Watch's public archive, and various news outlet databases.
Q: Did the CDC publish revisions to their modeling?
A: The CDC published updated modeling on its website, but the relationship between public messaging timelines and modeling timelines was never formally reconciled in released documents.
Q: How long did the restrictions ultimately last?
A: Many state-level restrictions remained in effect through mid-2021 (California through June 15, 2021; other states through various dates in 2021). The federal public health emergency remained in effect until May 11, 2023, making the actual duration approximately three years, not two weeks.
Related Reading
Explore more declassified pandemic policy documentation through these They Knew investigations:
- Operation Warp Speed: Pharmaceutical Contracts and Federal Authority
- CDC Guidance Evolution: A Timeline of Changes
- Government Messaging Precedents: Operation Mockingbird to Modern Communications
- FOIA and Institutional Transparency: How Documents Are Released
- Federal Health Agency Structure and Decision-Making Authority

