German company Chemie Grünenthal had reports of birth defects by 1959 but continued selling thalidomide for two more years, causing thousands of deformities.
“Thalidomide is completely safe for pregnant women and their unborn children”
From “crazy” to confirmed
The Claim Is Made
This is the moment they called it crazy.
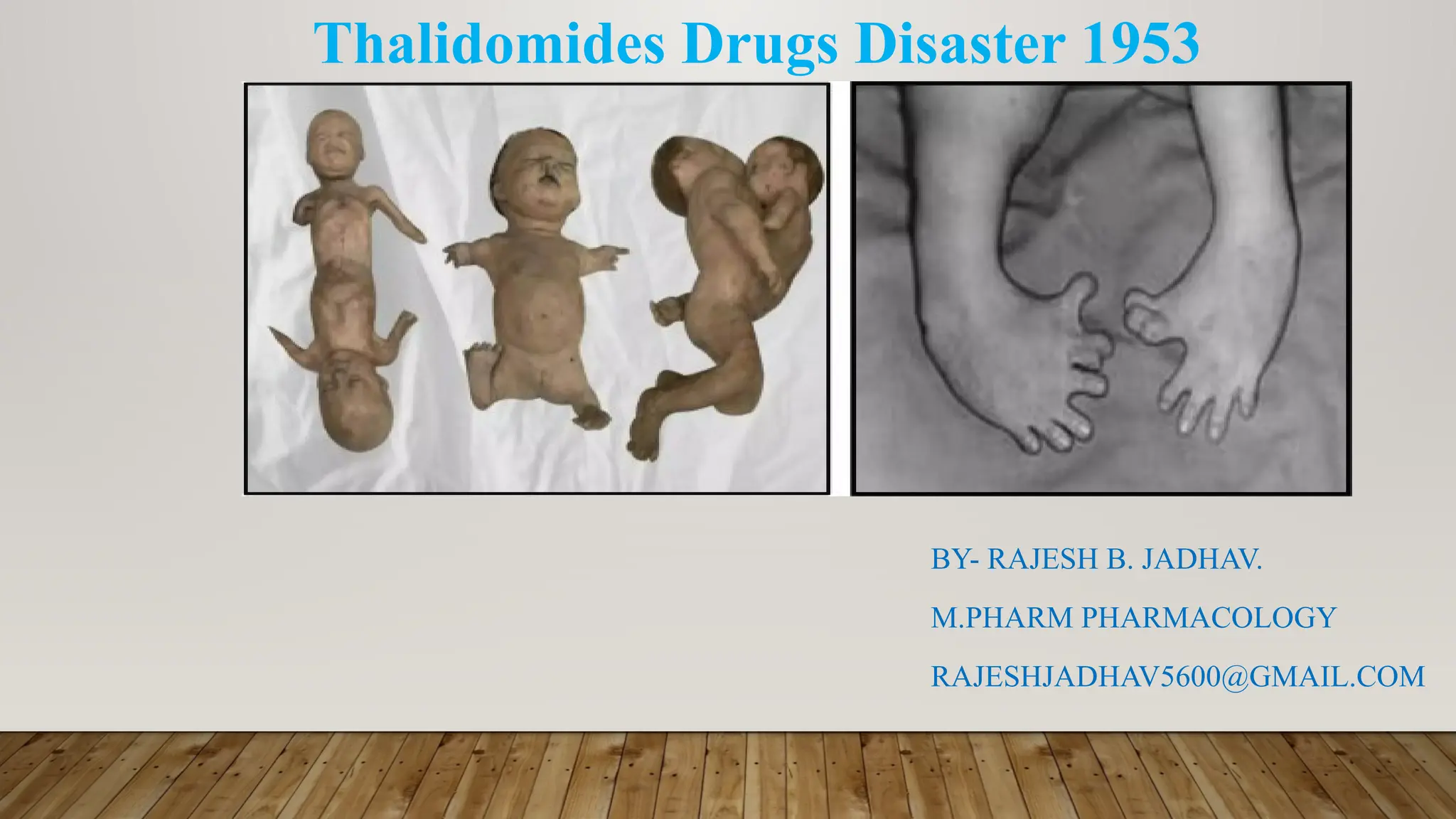
Between 1957 and 1961, a medication called thalidomide was prescribed to pregnant women across Europe as a treatment for morning sickness and insomnia. The drug was marketed as exceptionally safe, even for expectant mothers. What followed was one of the pharmaceutical industry's darkest chapters—and a textbook example of how institutions can suppress evidence to protect profits.
The tragedy didn't unfold because the dangers were unknown. German manufacturer Chemie Grünenthal had internal reports of severe birth defects associated with thalidomide as early as 1959. Children exposed to the drug in the womb were born with phocomelia—a rare condition where limbs failed to develop properly, leaving some with flipper-like appendages where arms and legs should have been. Other infants suffered damage to their eyes, ears, and internal organs.
Yet Chemie Grünenthal continued selling the drug. For two more years, while company executives possessed documented evidence of harm, the medication remained available across Europe. The official position from the company and many medical authorities was reassuring: thalidomide was safe. Regulatory bodies largely accepted this claim without demanding the rigorous investigation the evidence warranted.
The dismissal of warnings wasn't accidental. According to documentation examined in medical journals including the BMJ, internal communications at Chemie Grünenthal show that reports of birth defects were either downplayed or compartmentalized away from decision-makers in positions to halt production. When concerned physicians raised alarms, they were often met with corporate responses that questioned the reliability of their observations rather than acknowledging the accumulating evidence.
The proof of the claim came too late for thousands of families. By the time thalidomide was withdrawn from most European markets in late 1961 and 1962, approximately 10,000 children had been born with severe deformities. Historical analysis and retrospective examination of company records, as detailed in BMJ research, confirmed what the internal reports from 1959 onward had already suggested: the company knew, and it failed to act.
What makes this case essential reading today is not merely the tragedy itself, but what it reveals about institutional incentives. Chemie Grünenthal faced no criminal charges. The company paid settlements, yes, but the legal consequences were remarkably limited compared to the scale of harm caused. This disparity mattered. It demonstrated that suppressing safety information, while morally indefensible, could be treated as a manageable business risk.
The thalidomide tragedy reshaped pharmaceutical regulation in many countries. Stricter testing requirements and better oversight mechanisms emerged in its wake. But the underlying question persists: how many times since have warnings been minimized, delayed, or compartmentalized within companies in ways we haven't yet discovered?
This case matters because it established a documented pattern. Pharmaceutical companies possess safety information before regulators and the public do. The incentive structure—where continued sales generate revenue while disclosure generates liability—creates a built-in pressure toward silence. Thalidomide proved this isn't theoretical. It happened, with devastating consequences that could have been prevented if evidence had moved from internal reports to public knowledge faster.
Public trust in institutions depends on transparency. When that trust is violated, especially in healthcare, the damage extends far beyond those immediately harmed.
Get the 5 biggest receipts every week, straight to your inbox — plus an exclusive PDF: The Top 10 Conspiracy Theories Proven True in 2025-2026. No spam. No agenda. Just the papers they couldn't hide.
You just read "Thalidomide was known to cause birth defects but kept on Eur…". We send ones like this every week.
No one's said anything yet. Be the first to drop your take.





