Pfizer, Merck, J&J, Purdue — the receipts pharma wishes you'd stop reading.
Every blockbuster drug has a court filing under it. This timeline collects the documented cover-ups, suppressed trial data, FDA fights and settlements that turned 'conspiracy theory' into 'class action'.

Thalidomide was marketed in 46 countries for morning sickness without ever being tested on pregnant women. When German pediatrician Dr. Widukind Lenz identified the drug as causing birth defects, Grünenthal threatened him with legal action. The company bribed doctors, pressured medical journals to delay critical publications, and continued selling the drug. Over 10,000 babies were born with severe deformities; 40% died. Grünenthal didn't apologize until 2012 — over 50 years later.

Chemie Grunenthal marketed thalidomide as a safe sedative for pregnant women in the late 1950s despite having reports of severe nerve damage (peripheral neuropathy) and insufficient safety testing. The drug caused an estimated 10,000+ children to be born with severe limb deformities. Dr. Frances Kelsey of the FDA blocked its approval in the US, saving thousands. Internal documents showed the company received adverse reports but suppressed them for years.
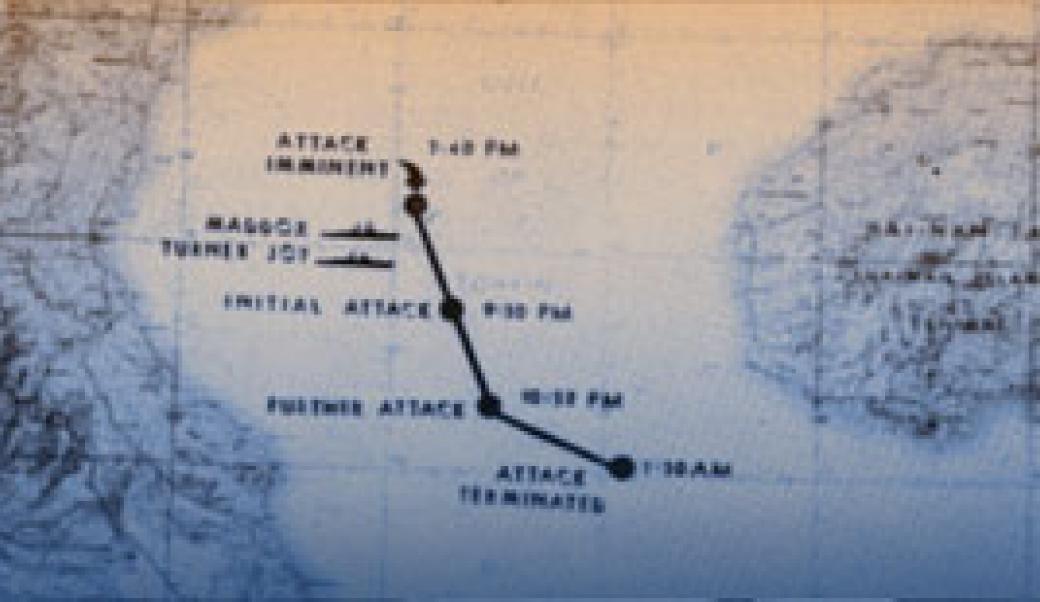
Declassified NSA documents from 2005 revealed that the second Gulf of Tonkin attack on August 4, 1964 never happened. NSA analysts 'made SIGINT fit the claim' of a North Vietnamese attack, deliberately presenting only information supporting the attack narrative to President Johnson. Congress passed the Gulf of Tonkin Resolution based on this fabricated evidence, escalating to full-scale war.
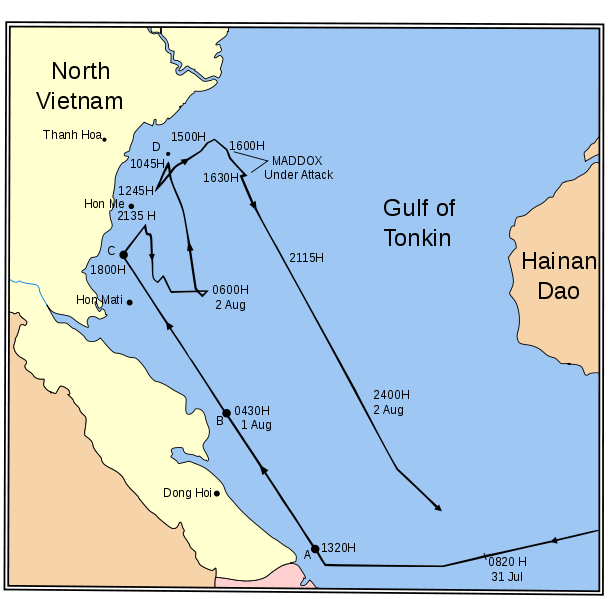
On August 4, 1964, the USS Maddox reportedly came under a second attack by North Vietnamese torpedo boats. President Johnson used this to push the Gulf of Tonkin Resolution through Congress, giving him unlimited military authority. NSA documents declassified in 2005 confirmed the agency deliberately manipulated signals intelligence to fabricate the second attack. Secretary McNamara later admitted the August 4th incident did not occur. The resolution led to the deaths of 58,000 Americans and millions of Vietnamese.
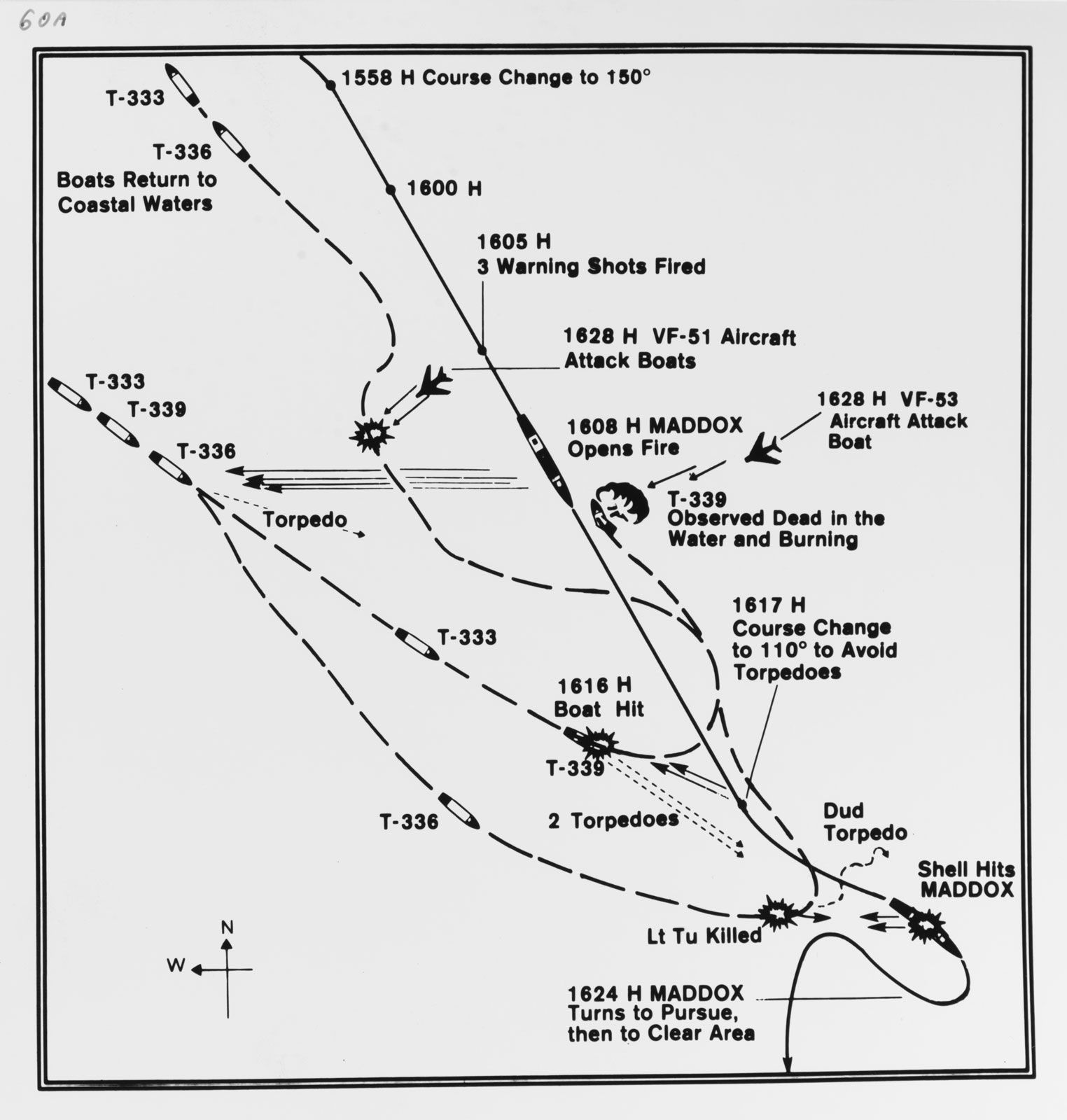
NSA documents revealed the second Gulf of Tonkin attack never occurred. Johnson administration used false reports of North Vietnamese attacks to obtain congressional authorization for massive military escalation in Vietnam.

Aspartame had a troubled FDA approval. A 1977 FDA task force found 'serious deficiencies' in safety studies by G.D. Searle. Commissioner Arthur Hull Hayes Jr. approved it in 1981, then left the FDA and joined Searle's PR firm. In 2023, the WHO's IARC classified aspartame as 'possibly carcinogenic' (Group 2B). Industry-funded studies consistently find it safe while independent studies more often identify concerns — a familiar pattern of funding bias.

The FDA placed a stay on aspartame approval in 1975 after discovering 'serious shortcomings' in all 15 long-term studies submitted by G.D. Searle. A grand jury investigation was requested but the US Attorney withdrew after considering a job at Searle's law firm. When Rumsfeld (Searle's CEO) helped elect Reagan, the new FDA Commissioner Arthur Hayes approved aspartame within months — against the conclusions of FDA toxicologists and an independent Board of Inquiry. Hayes then left the FDA to join Searle's PR firm.

When Bayer's Cutter Laboratories discovered their Factor VIII blood products were contaminated with HIV, they continued selling old unheated stock to markets in Asia and Latin America for over a year after introducing a heat-treated version in the US. Internal memos from 1983 acknowledged 'strong evidence' AIDS was transmitted through plasma products. An estimated 10,000+ hemophilia patients worldwide were infected with HIV. Bayer paid $600 million+ in settlements.

Since the 1970s, Dr. Stanislaw Burzynski has claimed his antineoplaston therapy can treat certain cancers. The FDA and Texas Medical Board repeatedly tried to shut him down, bringing criminal charges in the 1990s. He was acquitted of all 75 counts in 1997. Some patients reported results. However, no randomized controlled trials have proven efficacy, and the FDA cited numerous protocol violations. The case represents the tension between innovation persecution and the need for rigorous evidence.
Internal documents revealed Merck's marketing team developed the ADVANTAGE strategy to skew clinical trial results, comparing Vioxx to naproxen rather than a placebo. In 16 of 20 published papers, Merck employees were initially listed as lead authors before being swapped with outside academics. After 80 million patients took the drug, Merck withdrew it in 2004. The Lancet estimated 88,000-140,000 heart attacks in the US alone.
FDA inspection reports showed Abbott continued selling HIV blood screening tests with known defects that missed infections, risking blood supply contamination.
Merck's blockbuster painkiller Vioxx (rofecoxib) was linked to heart attacks and strokes. FDA whistleblower Dr. David Graham estimated Vioxx caused 88,000-140,000 excess heart attacks in the US, with an estimated 60,000 deaths. Internal emails showed Merck scientists identified the cardiovascular risk years before the 2004 withdrawal but the company minimized and concealed the data. Merck paid $4.85 billion to settle 27,000 lawsuits.
Pfizer secretly tested experimental antibiotic Trovan on 200 children during 1996 meningitis epidemic without proper consent. Eleven died, many suffered brain damage, but company claimed it was humanitarian aid.

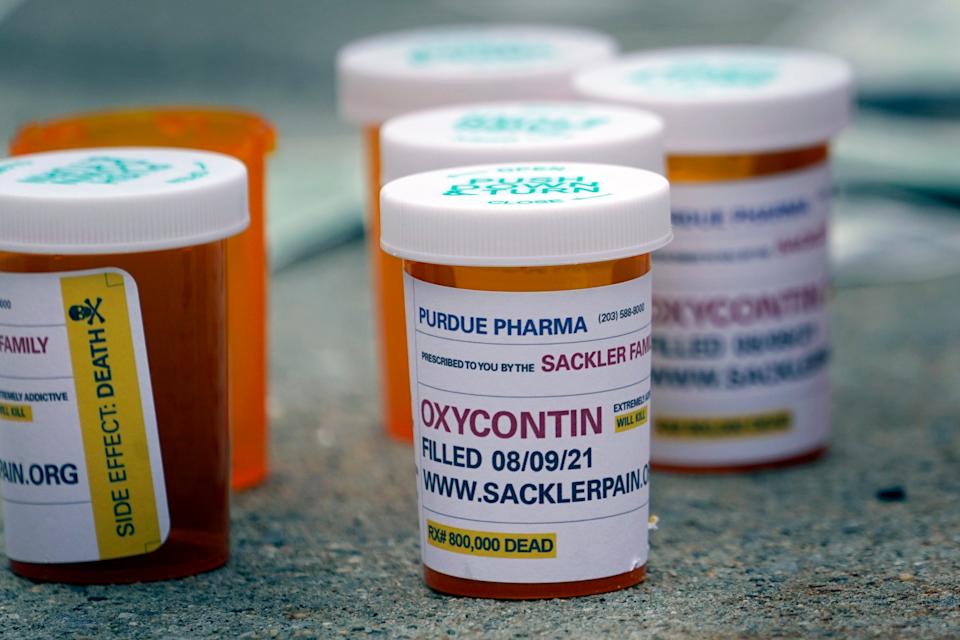
At OxyContin's 1996 launch, Richard Sackler predicted it 'will be followed by a blizzard of prescriptions that will bury the competition.' Purdue instructed reps to tell doctors OxyContin was not addictive, and that only those with an 'addictive personality' became addicts. When the epidemic emerged, Sackler devised a strategy to blame the victims. In 2007, a Purdue affiliate pleaded guilty to felony misbranding. Over 20 million documents were ordered disclosed. The Sacklers agreed to pay $4.325 billion while having extracted billions beforehand.

FDA documents show Bayer continued selling Factor VIII blood products contaminated with HIV and hepatitis to overseas markets after pulling them from the US in 1984.
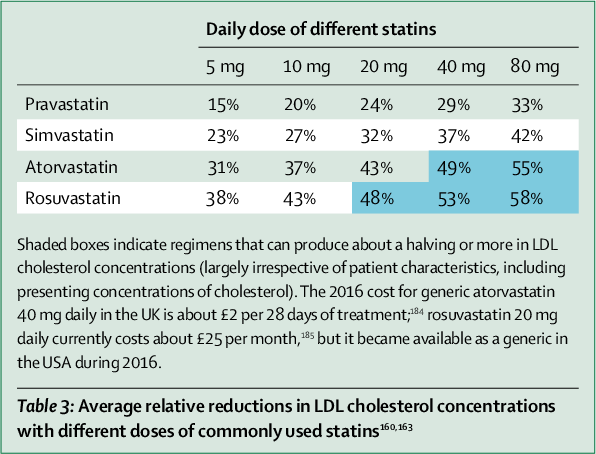
Statin drugs generated an estimated $1 trillion in cumulative sales, yet raw data on efficacy and safety have never been subjected to independent scrutiny. Less biased research shows guidelines underestimate adverse reactions by a factor of 10-100x. Common effects include muscle pain, cognitive decline, and increased diabetes risk. Hundreds of people need treatment for years for a single person to benefit. Critics argue the cholesterol hypothesis itself was driven by industry funding rather than evidence.

FDA scientists found SSRIs doubled suicide risk in children but agency suppressed findings until whistleblower leaked internal documents in 2004.
Internal emails showed Merck scientists knew Vioxx increased heart attack risk by 2000 but continued marketing it until 2004 withdrawal.
Internal Merck emails showed executives knew Vioxx doubled heart attack risk but suppressed studies and intimidated researchers. The company withdrew Vioxx only after external studies proved cardiovascular dangers.
Internal Merck documents showed the company knew Vioxx increased heart attack risk by 2000 but continued marketing until 2004. FDA emails revealed regulatory capture concerns.
Internal Merck emails showed executives knew Vioxx doubled heart attack risk as early as 1997. The company suppressed studies and attacked researchers who questioned its safety until 2004 withdrawal.
Internal documents showed Purdue executives knew OxyContin was highly addictive but trained sales reps to claim 12-hour dosing reduced addiction risk compared to other opioids.
Internal Purdue documents showed executives knew OxyContin caused addiction and overdoses while marketing it to doctors as having low abuse potential.
Court filings revealed Purdue executives received reports of widespread OxyContin abuse and addiction by 2000 but continued aggressive marketing claiming low addiction potential until 2007.

Court documents revealed Pfizer tested untested Trovan on 200 children in 1996 without proper consent, causing deaths and disabilities while effective treatment existed nearby.

A BMJ study examined 55 FDA staff who conducted drug reviews and found 15 of 26 who left the agency later worked for the biopharmaceutical industry. One reviewer who led FDA analysis of AstraZeneca drug reviews joined AstraZeneca as a director and was representing them before her former colleagues within a year. Four of the last five FDA Commissioners left to join pharmaceutical companies or their boards. A Stanford Law paper calls it 'one persistent source of undue influence at the FDA.'

Internal Purdue documents showed executives knew OxyContin's 12-hour relief claims were false and the drug was highly addictive. They aggressively marketed to doctors while concealing addiction data and blame-shifting to patients.

A Reuters investigation in December 2018 revealed that from at least 1971 to the early 2000s, Johnson & Johnson's talc and finished powders tested positive for asbestos. Traces were first detected in the late 1950s. J&J didn't tell the FDA that at least three tests from 1972 to 1975 found asbestos — one at levels reported as 'rather high.' Company executives, mine managers, scientists, doctors, and lawyers all knew about the contamination. They also successfully influenced regulators' plans to limit asbestos in cosmetic talc. J&J stock dropped 10% on the news — its worst loss in two decades.

The Sackler family, owners of Purdue Pharma, and consulting firm McKinsey & Company worked together to aggressively market OxyContin while knowing the drug was causing addiction and death. McKinsey proposed plans to 'turbocharge' sales and suggested Purdue pay rebates to pharmacies for OxyContin overdoses. Internal emails showed the Sacklers were directly involved in marketing decisions. The opioid crisis has killed over 500,000 Americans. Purdue pled guilty to felony charges; McKinsey paid $600 million in settlements.

Science: 9/10 commissioners to pharma. 57% of reviewers to companies they regulated. Companies hire staffers who managed their drug reviews.

A Reuters investigation revealed that J&J knew for decades that asbestos contaminated its talc-based baby powder. Internal documents dating to the 1970s showed the company was aware of the contamination and the cancer risk but chose not to disclose it. The company faced over 40,000 lawsuits linking its talc products to ovarian cancer and mesothelioma. J&J eventually discontinued talc-based baby powder in 2023 and proposed an $8.9 billion settlement.

Internal J&J memos from 1971-2000s revealed company executives knew talc in baby powder contained asbestos. They concealed test results showing contamination while marketing products as pure and safe for families.

McKinsey & Company worked on 37 FDA contracts while simultaneously consulting for Purdue Pharma on strategies to boost OxyContin sales, a massive conflict of interest they never disclosed to the FDA. McKinsey used its government work to solicit business from opioid manufacturers and tried to influence Trump HHS Secretary Alex Azar for private opioid clients. In July 2018, senior partners discussed destroying their Purdue documents, with one emailing himself a note to 'delete old pur documents from laptop.' McKinsey paid $650 million to resolve criminal and civil investigations.
Court documents revealed Sackler family members personally directed Purdue's aggressive opioid marketing strategies while privately acknowledging addiction risks.

In early 2021, the idea of vaccine passports was ridiculed as conspiracy thinking. By late 2021, Israel implemented the Green Pass, the EU launched digital COVID certificates, Canada mandated them across all provinces, and Boris Johnson reversed his opposition to implement them in the UK. PM Trudeau called them 'divisive' before mandating them.
Brook Jackson, a regional director at Ventavia Research Group (a Pfizer subcontractor), filed a complaint with the FDA in September 2020 reporting data integrity issues at her trial site including falsified data, unblinded patients, inadequately trained vaccinators, and failure to follow up on adverse events. The BMJ investigated and corroborated her claims. The FDA did not inspect Ventavia's site. Her False Claims Act lawsuit was dismissed on procedural grounds.

Brook Jackson, regional director at Ventavia Research Group, witnessed falsified data, unblinded patients, poorly trained vaccinators, and delayed adverse event follow-ups during Pfizer's pivotal COVID-19 vaccine trial. She reported to the FDA and was fired within hours. The FDA never inspected the Ventavia trial sites. Jackson filed a False Claims Act lawsuit with 400 pages of evidence. The DOJ declined to intervene, arguing it wasn't in the government's interest.
Six of seven 2021 meta-analyses of ivermectin RCTs found notable reductions in COVID fatalities, with a mean 31% reduction in mortality. The drug won the 2015 Nobel Prize for its creators. Yet media universally mocked it as 'horse dewormer,' social media censored discussion, and pharmacies refused to fill prescriptions. The FDA tweeted 'You are not a horse. Stop it.' Later studies showed mixed results, but the suppression of early investigation and the campaign of ridicule remain unprecedented in medical history.
A Lancet study across 65 countries found natural immunity provided 88%+ protection against severe disease at 10 months post-infection. An Israeli study showed natural immunity was stronger than two-dose Pfizer vaccination against Delta. Despite this evidence, the CDC refused to recognize natural immunity in its policies, and the Washington Post noted 'policymakers seem afraid to say so.'

A 2021 Congressional investigation found that popular baby food brands including Gerber, Beech-Nut, and HappyBABY had 'dangerous' levels of toxic heavy metals. Internal company documents showed manufacturers were aware of contamination but routinely ignored their own internal standards and failed to test finished products. Companies sold products with arsenic levels up to 91x the FDA's limit for bottled water. The FDA's 'Closer to Zero' initiative was launched only after congressional pressure.

Brook Jackson: falsified data, unqualified staff. Reported to FDA, fired. FDA inspected 9/153 sites - none hers.

Gottlieb: FDA May 2017-April 2019, Pfizer board June 2019. Twitter Files: flagged tweets as Pfizer child vaccine pending. Twitter found no violation but labeled anyway.
Genomics researcher Kevin McKernan identified SV40 promoter-enhancer sequences and residual plasmid DNA in Pfizer and Moderna mRNA vaccines. A peer-reviewed study in Autoimmunity found DNA exceeded FDA regulatory limits by 36-153x for Pfizer and 112-627x for Moderna using fluorometry. Two of six Pfizer lots exceeded regulatory limits for SV40 sequences specifically. Regulators have not mandated recalls despite the findings.
GLP-1 receptor agonists like Ozempic (semaglutide) generated $18 billion in 2023 sales while post-market adverse event reports accelerated. The FDA received thousands of reports of gastroparesis (stomach paralysis), severe pancreatitis, bowel obstruction, and suicidal ideation — side effects that were either underrepresented or absent from clinical trial data. European regulators launched investigations into thyroid cancer links and suicidal thoughts. Novo Nordisk's stock reached record highs while patient advocacy groups argued the company minimized risk disclosures.

University of Toronto study in the Journal of Law, Medicine & Ethics analyzed 20+ years of OECD data. Pfizer, J&J, Eli Lilly, BMS, AstraZeneca, and GSK all ran systematic bribery via shell companies. $1.1B in sanctions. Internal compliance warnings were ignored.

Lawsuits allege Pfizer had studies since 1983 linking Depo-Provera to meningioma brain tumors. Women on the shot 1+ year are 5x more likely to develop brain tumors. Pfizer only added a warning in December 2025 — 42 years later. 3,099 lawsuits filed.

At sentencing on April 22, 2026, Purdue Pharma forfeited $225M to the DOJ and the Sackler family agreed to pay up to $7 billion in civil settlements. No member of the Sackler family will face criminal prosecution. Survivors testified the plea deal locks in permanent impunity.

KC Pharmaceuticals issued a nationwide recall of over 3 million eye-drop bottles after Pseudomonas aeruginosa contamination was detected. The facility had received an FDA warning letter for sterility failures in 2023. Products continued to ship to CVS, Walgreens, and Kroger in the intervening period.

Pfizer and its generic subsidiary Greenstone agreed to pay $33 million as part of a $70.75 million multi-defendant antitrust settlement over alleged bid-rigging and customer-allocation schemes in the U.S. generic drug market.
Every entry on this timeline started as a tip. If you have documentation, a court filing, a leaked memo or a screenshot — drop it.
+ Drop a Receipt